The "DysHema" team studies the molecular mechanisms that lead to malignant transformation in human hemopathies in order to identify new molecular targets and active molecules that will define innovative therapeutic strategies. We are particularly interested in Acute Myeloid Leukemia (AML) and T-Acute Lymphoblastic Leukemia (T-ALL) in children, two leukemias which, despite decades of improved treatments, still have a poor outcome for relapsed patients.
The team conducts fundamental and translational research in partnership with the pediatric oncology department of the Nice University Hospital. In addition to our long experience in biochemistry and cell and molecular biology, we have set up collaborations with chemists and biochemists in cryo-EM and developed bioinformatics approaches to achieve our objectives:
- to identify and study deregulation events involved in addiction and molecular resistance
- to analyze the pathological properties of leukemic stem cells that generate leukemia, maintain the disease, and trigger therapeutic failure and relapse.


Projects

JF. PEYRONResearch Director
Mail Jean-Francois.PEYRON@univ-cotedazur.fr
Cancer cells reprogram their metabolic pathways to optimize nutrient and energy supplies for exacerbated proliferation, escape from apoptosis, and resistance to treatment. Targeting their deregulated metabolism could lead to new antitumor strategies.
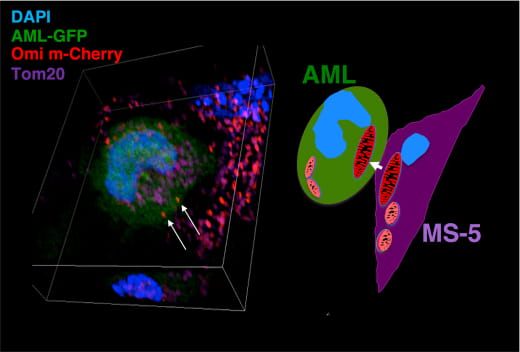
3D confocal microscopy reconstruction of an AML-GFP coculture with MS5-Omi-MCherry stromal cells.
Mitochondria staining with anti-Tom20.
The bone marrow microenvironment (BMME) is a complex ecosystem that instructs and protects hematopoietic stem cells (HSCs) and their malignant counterparts, leukemia stem cells/leukemia initiating cells (LSCs/ICs). Among the different functional exchanges that take place in the BMME, organelle transfer is now recognized as an important mode of intercellular communication, along with the role of adhesion molecules, secreted cytokines, and (onco)metabolites.
Using a coculture model mimicking the hematopoietic niche, we demonstrated that Acute Myeloid Leukemia (AML) cells subjected to chemotherapeutic stress trigger a unidirectional transfer of active mitochondria from surrounding bone marrow stromal cells. This uptake of mitochondria increases their mass and mitochondrial fitness, enhances their long-term leukemic culture initiation potential and their resistance to chemotherapy.
Targeting the mitochondrial transfer that provides a clear survival advantage to leukemic cells could represent an additional new strategy to interfere with the supportive actions of the BMME during leukemia progression and relapse.
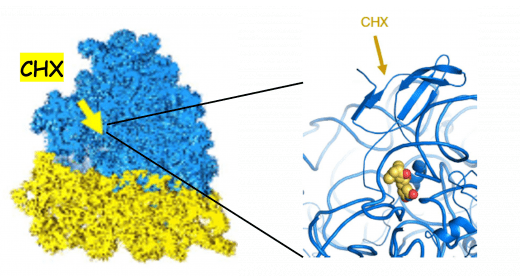
First cryo-EM structure of a CHX/80S complex.
Mean resolution : 3.6 Å
Based on the knowledge of high-resolution atomic interactions of the protein synthesis inhibitor cycloheximide (CHX) with the human 80S ribosome, we created a consortium in 2017 consisting of three teams: Bruno Klaholz's biochemistry laboratory (IGBMC, Illkirch) for cryo-EM structures, Jean-Yves Winum's laboratory (Institut des Biomolécules Max Mousseron, Montpellier) for organic and medicinal chemistry, and J-F Peyron's laboratory (C3M, Nice) for in vitro and in vivo functional characterization of anticancer molecules. We have developed a drug-design strategy using the knowledge of the CHX-80S structure to synthesize more efficient and selective CHX derivatives with antileukemic properties. These new derivatives are currently being characterized in vitro: analysis of antileukemic effects against human and murine L/LAL-T lines and toxicity against healthy cells. A selection of the most effective and least toxic molecules will be tested in a LAL-T-PDX (Patient-derived Tumor Xenografts) model.
This project is supported by InCa (PLBIO 2016 and 2021).

V. ImbertResearcher
Mail veronique.imbert@univ-cotedazur.fr

M. NEBOUTResearch engineer
Mail nebout@unice.fr

D. MARYResearcher
Mail Didier.MARY@univ-cotedazur.fr
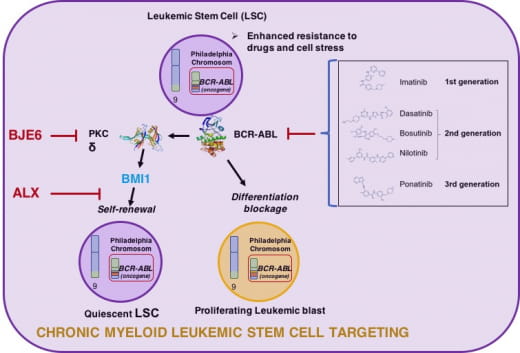
The silencing of BMI1 is deleterious in a large number of cancers, but the possibility of inducing such a response with pharmacological means remains very limited. To date, there is no molecule that has passed the clinical trial phases to directly target BMI1.
In a core program, we developed a strategy to act "downstream" of the polycomb protein Bmi1, crucial for the self-renewal function of normal or cancerous stem cells, by a bioinformatics approach using the Connectivity Map (cmap) database. This allowed us to identify molecules belonging to the bis-biguanide family, some of which were found to have a strong antitumor potential in Chronic Myeloid Leukemia CML cell lines. More interestingly, these bis-biguanides potentiate the effects of BCR-ABL inhibitors (ITKs) on both sensitive and resistant cells.
In a second core program, we have shown the involvement of PKC delta in the signaling pathway between BCR-ABL and the regulation of Bmi1 gene expression. In a collaboration with an American team specialized in the synthesis of kinase inhibitors, we used a specific inhibitor of PKCd (BJE6) and showed its ability to potentiate the effect of TKIs and target the leukemic stem cell compartment in stromal cell coculture systems.
This project was funded by FiLMC-Pfizer and Cancéropôle PACA.

M. NEBOUTResearch engineer
Mail nebout@unice.fr

V. ImbertResearcher
Mail veronique.imbert@univ-cotedazur.fr
NF-kappaB is a transcription factor that controls multiple cellular processes essential for immunity, inflammation, tumor development, chemoresistance and radioresistance. Our team has contributed significantly to the identification of this protumor role of NF-kappaB.
We have an in vivo model representative of human T-cell Lymphoma and Acute Lymphoblastic Leukemia (LL-T/LAL-T), generated by deletion of the tumor suppressor gene PTEN specifically in T cells (tPTEN-/-). The absence of PTEN leads to abnormal constitutive activation of the PI3K-Akt-mTOR axis, a feature found with high frequency in human pathology. During the characterization of the deregulated signaling pathways in this tPTEN-/- model, we observed a constitutive activation of NF-kappaB. To better understand its role in lymphoma initiation and progression, we adopted a genetic approach. Thus, the invalidation of NF-kappaB in the tPTEN-/- model allowed us to observe a tumor suppressor role for NF-kappaB in lymphoma. We are currently studying this role at different levels: antitumor response, gene stability.
This project is supported by the ARC Foundation.

M. NEBOUTResearch engineer
Mail nebout@unice.fr
Publications
Focus
Identifying Candidate Gene Drivers Associated with Relapse in Pediatric T-Cell Acute Lymphoblastic Leukemia Using a Gene Co-Expression Network Approach.Authors Kypraios A, Bennour J, Imbert V, David L, Calvo J, Pflumio F, Bonnet R, Couralet M, Magnone V, Lebrigand K, Barbry P, Rohrlich PS, Peyron JF
Cancers May 2024
Structure-function insights reveal the human ribosome as a cancer target for antibiotics.Authors Myasnikov AG, Kundhavai Natchiar S, Nebout M, Hazemann I, Imbert V, Khatter H, Peyron JF, Klaholz BP
Nature communications Sep 2016
Other recent publications
Identifying Candidate Gene Drivers Associated with Relapse in Pediatric T-Cell Acute Lymphoblastic Leukemia Using a Gene Co-Expression Network Approach.Authors Kypraios A, Bennour J, Imbert V, David L, Calvo J, Pflumio F, Bonnet R, Couralet M, Magnone V, Lebrigand K, Barbry P, Rohrlich PS, Peyron JF
Cancers May 2024
Repurposing the Bis-Biguanide Alexidine in Combination with Tyrosine Kinase Inhibitors to Eliminate Leukemic Stem/Progenitor Cells in Chronic Myeloid Leukemia.Authors Muselli F, Mourgues L, Rochet N, Nebout M, Guerci A, Verhoeyen E, Krug A, Legros L, Peyron JF, Mary D
Cancers Feb 2023
Reduced-toxicity myeloablative conditioning regimen using fludarabine and full doses of intravenous busulfan in pediatric patients not eligible for standard myeloablative conditioning regimens: Results of a multicenter prospective phase 2 trial.Authors Rialland F, Grain A, Labopin M, Michel G, Gandemer V, Paillard C, Pochon C, Clement L, Brissot E, Jubert C, Sirvent A, Rohrlich PS, Plantaz D, Dalle JH, Mohty M
Bone marrow transplantation Aug 2022
Show all publicationsPatents
Co-inventors J. PEYRON
Co-inventors J. PEYRON
ARC Contributors' Day, May 2017

PRESS REVIEW





